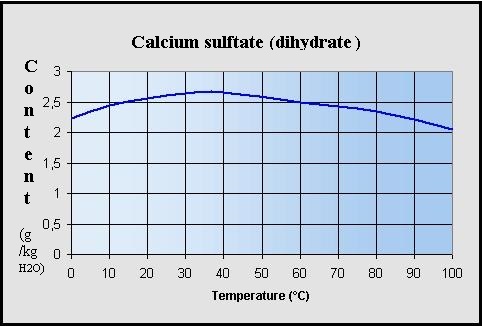
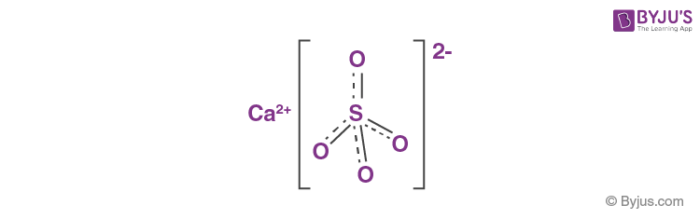
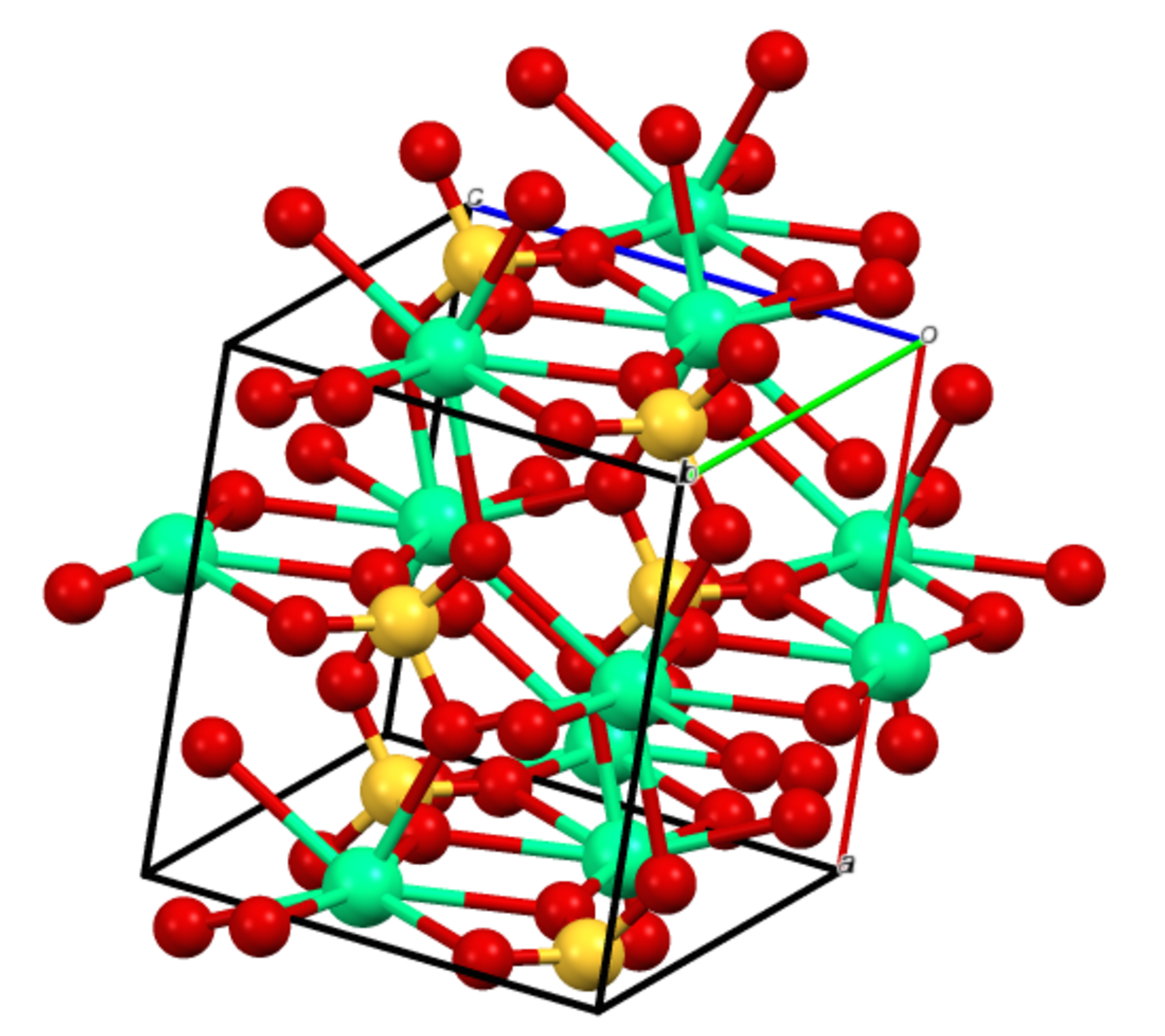
Thermodynamic Modeling of Calcium Sulfate Hydrates in the CaSO4–H2O System from 273.15 to 473.15 K with Extension to 548.15 K | Journal of Chemical & Engineering Data
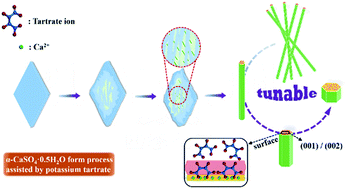
Aspect ratio-controlled preparation of α-CaSO4·0.5H2O from phosphogypsum in potassium tartrate aqueous solution - RSC Advances (RSC Publishing)

The effect of additives on the hydration of CaSO4·0.5H2O: A synchrotron X-ray diffraction study - ScienceDirect
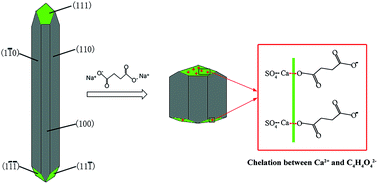
Synthesis of α-CaSO4·0.5H2O from flue gas desulfurization gypsum regulated by C4H4O4Na2·6H2O and NaCl in glycerol-water solution - RSC Advances (RSC Publishing)

Formation and Transformation of Five Different Phases in the CaSO4−H2O System: Crystal Structure of the Subhydrate β-CaSO4·0.5H2O and Soluble Anhydrite CaSO4 | Chemistry of Materials
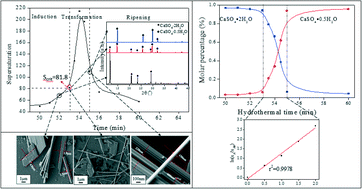
Supersaturation-induced hydrothermal formation of α-CaSO4·0.5H2O whiskers - CrystEngComm (RSC Publishing)
Force field for calcium sulfate minerals to predict structural, hydration, and interfacial properties

Influence of the Process Parameters on the Formation of CaSO4·0.5H2O Whiskers – topic of research paper in Nano-technology. Download scholarly article PDF and read for free on CyberLeninka open science hub.